 Topic 8: Metabolism, cell respiration, and photosynthesis (Higher level)
Topic 8: Metabolism, cell respiration, and photosynthesis (Higher level)
This is an introduction to the HL Metabolism topic. It lists understandings and skills expected for Topic 7 including; extra details of enzymes, enzyme inhibitors, cell respiration and photosynthesis. Helpful for revision.
Detailed revision notes, activities and questions can be found on each of the sub-topic pages
- 8.1 Metabolism
- 8.2 Cell respiration
- 8.3 Photosynthesis
This pages gives outline details of the content of the topic together with essential questions and student skills and applications.
8.1 Metabolism
Metabolic pathways
- A chain or a cycle of enzyme-catalysed reactions makes a metabolic pathway.
- Enzymes catalyse chemical reactions by lowering the activation energy.
Competitive inhibitors
- Competitive inhibitors for example in medicine - ethanol and Fomepizole used as competitive inhibitors for antifreeze poisoning.
Non-competitive inhibitors
- Non-competitive inhibitors. E.g. Drugs for treatment of malaria.
- End-product inhibition can control metabolic pathways - e.g. In the pathway that converts threonine to isoleucine.
Skills
- The use of databases to identify potential new anti-malarial drugs. In the ER role play: students will have seen this
- In the Enzyme inhibition experiment students will calculate and plot rates of reaction from raw experimental results.
- They will also distinguish different types of inhibition from graphs at specified substrate concentration.
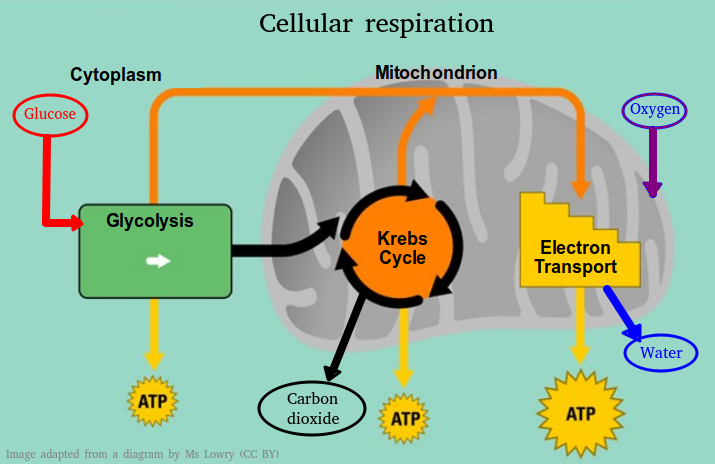
8.2 Respiration
Introduction to reactions in respiration
- Cell respiration involves:
- the oxidation and reduction of electron carriers.
- the phosphorylation of molecules which makes them less stable.
- decarboxylation removes a carbon atom from a molecule
Glycolysis & the Krebs cycle
- Glycolysis is the conversion of glucose to pyruvate in the cytoplasm giving a small net gain of ATP(without the use of oxygen.)
- Aerobic cell respiration converts the pyruvate by decarboxylation and oxidation, into an acetyl compound.
- In the link reaction acetyl is attached to coenzyme A to form acetyl coenzyme A.
- In the Krebs cycle,
- the oxidation of acetyl groups is coupled to the reduction of hydrogen carriers, liberating carbon dioxide.
- reduced NAD and FAD (NADH+H+ and FADH+) carry energy released by oxidation reactions to the cristae of the mitochondria.
The electron transport chain & Mitochondrion structure.
- the electron transport chain in the membrane of the cristae is coupled to proton pumping.
- oxygen is the final electron acceptor.
- In chemiosmosis protons diffuse through ATP synthase to generate ATP.
- Oxygen binds with the free protons to maintain the hydrogen gradient and resulting in the formation of water.
- The structure of the mitochondrion is adapted its function. Annotate a diagram of a mitochondrion to show these adaptations
Skills
- Active mitochondria have been studies using electron tomography - images.
- students will be able to identify where decarboxylation and oxidation reactions occur in diagrams of the pathways of aerobic respiration.
- Guidance:
- The names of the intermediate compounds in gylcolysis and the Krebs cycle are not required.
8.3 Photosynthesis
Light-dependent reactions
- Happen in the thylakoid membranes and in the space inside them.
- Reduced NADP (NADPH2) and ATP are produced in the light-dependent reactions.
- Light absorbed by photosystems generates excited electrons.
- Photolysis of water generates electrons for use in the light-dependent reactions.
- Excited electrons are transfered between carriers in the thylakoid membranes.
- Excited electrons from Photosystem II are used to generate a proton gradient.
- ATP synthase in the thylakoids generates ATP using the proton gradient.
- Excited electrons from Photosystem I are used to reduce NADP to NADPH2.
Light-independent reactions
- Take place in the stroma.
- RuBP carboxylase enzyme catalyses the carboxylation of ribulose bisphosphate (RuBP).
- Glycerate 3-phosphate is reduced to triose phosphate using "reduced NADP" (NADPH2) and ATP.
- Triose phosphate is used to regenerate RuBP and produce carbohydrates.
- Ribulose bisphosphate (RuBP) is regenerated using ATP.
Chloroplast structure
- The structure of the chloroplast is adapted to its function in photosynthesis.
Skills
- to recognise details of Calvin’s experiment to elucidate the carboxylation of RuBP.
- to be able to annotate a diagram showing the adaptations of a chloroplast to its function.

Metabolism 8.1 HL
This topic covers the way enzyme pathways form a cell's metabolism and specific examples of these pathways. The action of enzymes and their substrates is required, competitive and non-competitive inhibitors as well as calculating rate of reaction.

Cell respiration 8.2 HL
This topic looks at the details of glycolysis, the link reaction and the krebs cycle. The structure of mitochondria is related to its function and the amazing process of chemiosmosis is outlined leading to the production of water and ATP.

Photosynthesis 8.3 HL
This topic covers the tricky details of the light dependent and light independent reaction of photosynthesis. This includes photolysis and oxidative phosphorylation as well as chemiosmosis where ATPsynthase enzyme generates ATP.

 Twitter
Twitter  Facebook
Facebook  LinkedIn
LinkedIn